Microfluidics for Energy & Environmental Applications
Using microfluidics techniques combined with optical diagnosis, we aim at exploring multiphase flow dynamics at various scales in energy and environmental-related applications. One example is for enhanced oil recovery using CO2 flooding.
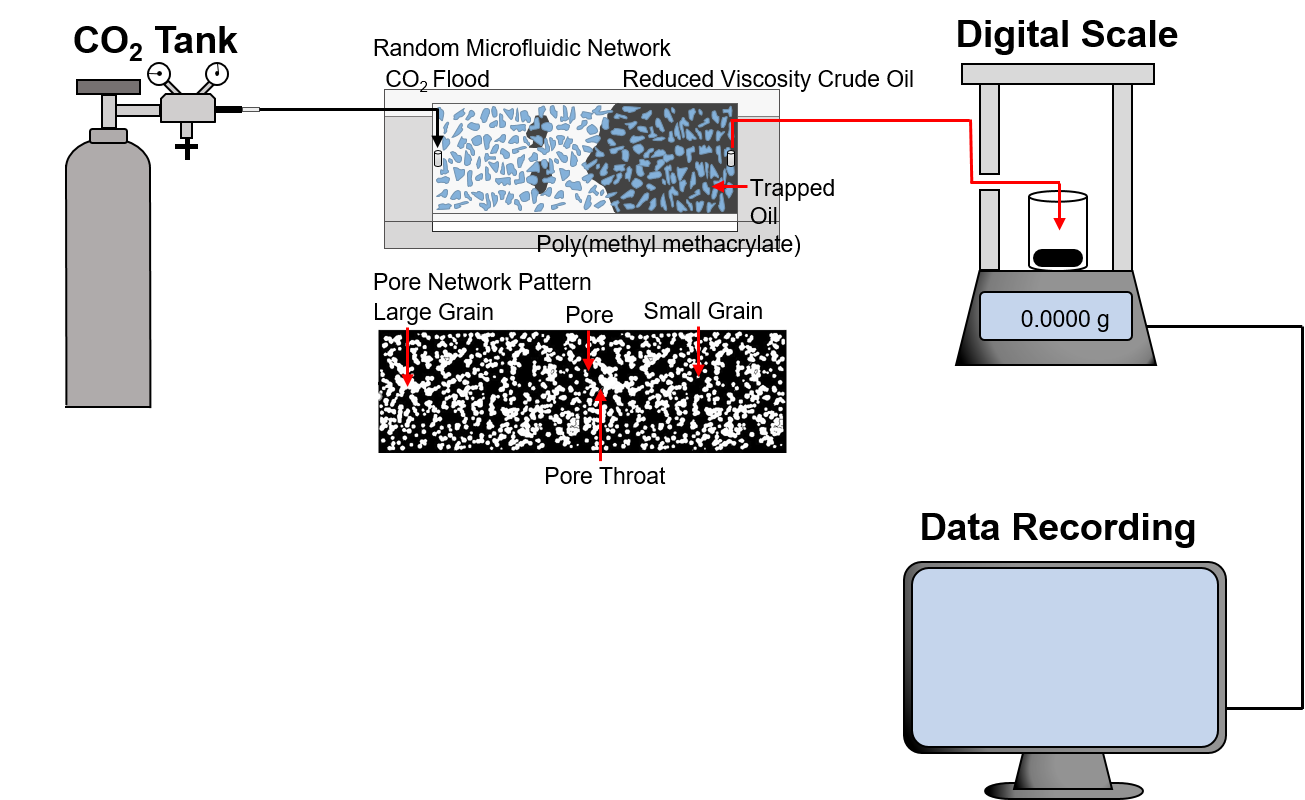
CO2-Flooded Enhanced Oil Recovery
Chemical enhanced oil recovery (EOR) is a successful method for increasing crude oil recovery. However, chemicals commonly used for enhanced oil recovery operations possess adverse biological impacts. To meet the legislative requirement and environmental protection demands, the performance of a highly biodegradable nonionic surfactant derived from tannic acid, a possible alternative, was evaluated using a microfluidic technology for the replacement of chemically synthesis surfactant by green chemistry products. Aqueous microdroplets containing the surfactant in crude oils were used for measurements of interfacial tension (IFT) reduction. The degree of interfacial tension reduction by sodium dodecyl sulfate (SDS), one of the most popular conventional surfactants, was also quantified for performance comparison. The potential of the biosurfactant for IFT reduction of light crude oil was superior to that of SDS. To evaluate the feasibility of the biosurfactant in improvement of recovery efficiency, surfactant-assisted flooding was tested under a random microfluidic network at the optimal concentrations, and the results were in good agreement with IFT reduction tests. The utilization of the polymer in a biosurfactant synthesis process effectively enhanced high sweep efficiency by decreasing a viscous fingering effect. The biosurfactant proved to be adequate and can sufficiently alleviate environmental concerns adopted by chemical flooding EOR.
[Test Setup]
[Flooding Experimental Results]